
Dec 4, 2020
The feeling? Absolute terror! So many things I thought I could never do again. Walking a mile? It took everything I had to walk 10 feet! Walking on uneven ground? I”ll never be able to do that again! Stairs? Like Mountain climbing with only one leg. Even getting up from a sitting position was a monumental task. I had let my right knee deteriorate much too far. It was so bad that I was ready and denser-ate for a replacement.
My friend saw astonishing results from some of his Business Customers and recommended that I check out they Stem Cell procedure.
With an initial assessment by Dr. Sheinkop, the procedure was done a few weeks later. After the procedure, I walked out with no more pain than when I walked in. A bandage covering 2 small holes in my back, a Band Aid on my knee, and a custom-fit knee brace, to wear for four weeks.
Not hospital stay, no x-rays, no pain killers, no prescriptions, no unnecessary tests, and no depressing stay in a Rehab facility. And, no artificial device.
Progress was slow but steady – two steps forward, one step back.
The Stem Cell Facts
At first, stairs were near impossible. I had a stinging pain in the front of my knee, and a severe limp. After six months, my knee condition improved to about 60%, with therapy and home exercises. The stinging pain had diminished. Stairs were difficult but I could manage up and down while holding on. Limp had improved from severe to awkward.
At the one year mark, my knee was light, stronger, and more flexible – and the stinging pain was gone. Had slight pain while going down stairs, with an improved but still noticeable limp. My self-assessment at this time was 93% There was some anxiety. I read full healing takes about one year. I had no idea that my condition would continue to improve.
It improved dramatically, but not quickly. A year-and-a-half after the procedure, all pain, soreness, and stiffness was gone, my knee was strong and flexible. I did thinks I wasn’t sure I’d be able to do again – kneeling, crawling, walking, running, bicycle riding, jumping, climbing, golf, hockey – too many to mention. I was at 99% and it felt great! The remaining 1% was a slight, but still noticeable limp.
It took another six months for the limp to completely go away. This was two years after the procedure. 100% – back to normal!
For something that sounded too good to be true, the results are there. And, the cost? For me, it was less than the deductible I would have paid for a conventional operation.
One of the best decisions I ever made was to have this Stem Cell procedure done.
Find out if You’re a Candidate for Dr. Sheinkop’s Stem Cell Procedure
Tags: cellular therapy facts, patient testimonials, stem cell procedures, stem cell therapy

May 29, 2020
There are statistics and anecdotes that are used to review the outcomes of a particular cellular orthopedic treatment option and help determine what is best for a patient with arthritis. At times, it is difficult, and even impossible, to develop a sufficient number of patients with similar problems that might enable a statistical analysis. At that time, we have to base our recommendations on anecdotes; that is small numbers of patient responses. Two and a half years ago, a 67-year-old cycling enthusiast presented to our office with progressive pain and loss of motion owing to osteoarthritis of both knees. By the time he presented, the customary approaches to the arthritic knee had been exhausted including Physical Therapy, analgesics, non-steroidal anti-inflammatory medications, Hyaluronic Acid injections and arthroscopic “clean out.” Total Knee Replacements had been prescribed as the treatment option when all else had failed.
Patient Outcome Anecdotes
The patient had learned of my Cellular Orthopedic initiatives from his cycling group and sought consultation in our offices. The “team” of senior cyclists had experienced the recent loss of two team members because of their having experienced less than optimal outcomes following Total Knee Replacements. After our office assessment and review of images, I recommended a Bone Marrow Concentrate injection in both knees to help post postpone, perhaps avoid major surgery in the avid cyclist. I chose Bone Marrow Concentrate as the optimal means of delivering Adult Mesenchymal Stem Cells, Platelets, Precursor Cells and Growth Factor Proteins. The procedures were done on November 22nd of 2017. At this yearly follow up assessment, the patient reported that he has no pain, no limitation of function, requires no medications for arthritis and enjoys an unlimited weekly cycling routine approaching 300 miles per week.
A second example, or anecdote, is this senior athletic enthusiast, me. Many who know me are aware of my arthritic knees and hips for which I have had two rounds of biologic interventions in the past four years. Over the Memorial Day weekend, I cycled a combined 70 miles in Southwest Wisconsin, on hilly terrain, at times accompanied by my wife and twin grandsons; finished planting a large garden by hand; and helped clean our farmhouse, as we hosted and entertained our three grandchildren( ages 12, 10 and 10) with parents -the latter the most strenuous task of all.
While there is a medical cliché, “an anecdote does not a series make”, the vignettes above are real examples of what may be done for a symptomatic and functionally limiting arthritic joint. To learn more about orthobiologic options from proprietary Platelet Rich Plasma to stem cells and then some, call and schedule an appointment at (312) 475-1893. You may visit my website at www.sheinkopmd.com and watch my webinar.
Tags: arthritis pain, joint pain, patient outcomes, stem cell therapy, stem cell treatments

Jan 7, 2020
This excerpt is taken from the full article, which can be read here. Copyright © Mitchell B Sheinkop and Maxwell Dordevic,2019,
Abstract
This case report will introduce the concept of using an acellular mesenchymal stem cell (MSC) derived extracellular vesicle isolate product (EVIP) containing active growth factors (GFs) and exosomes to treat hip osteoarthritis (OA) as well as the rationale of why acellular may replace all current cellular biologic therapies both autogenous and allogeneic presently in use.
Excerpt
INTRODUCTION Hip osteoarthritis (OA) has demonstrated, in both cadaver and radiographic studies, to affect up to 55 million patients over the age of 60.1 Patients with hip OA have pain, crepitus, loss of motion, and decreased ability to weight bear or ambulate. Limiting the ability to ambulate severely impairs activities of daily living. The nonsurgical treatments for hip OA according to the American Academy of Orthopedic Surgeons (AAOS),include weight loss, gentle exercise, and the use of non-steroidal anti-inflammatory medications. The surgical treatment for hip OA is total hip arthroplasty (THA).2 The AAOS does not recommend hip arthroscopy or the use of any Hyaluronic Acid injections. Over the last few years, it has become increasingly understood by researchers and clinicians that the clinical efficacy of utilizing mesenchymal stem cells (MSCs) to treat osteoarthritis (OA) is not dependent on the cells differentiating into articular cartilage but entirely on their paracrine release of growth factors (GFs) and exosomes. Living MSCs are not required to accomplish the release of GFs and exosomes into an arthritic joint. This case report will introduce the concept of using an acellular MSC derived extracellular vesicle isolate product(EVIP) containing active growth factors and exosomes to treat hipOAas well as the rationale of why acellular may replace all current cellular biologic therapies both autogenous and allogeneic presently in use.
MATERIALS AND METHODS This is a case report of an EVIP injection for the treatment of hip osteoarthritis. OA is defined by swelling, pain, and stiffness in the hipjoint. Symptoms are typically worsened by weight bearing and ambulation.Radiographs and MRI scanning wereused tograde osteoarthritis of the hip joint from one to four using the Kellgen-Lawrence scale.12 The patient is a 63-year-old retired Chicago Fireman. He presented with increasing pain in the left groin and a progressive loss of ability to continue his daily health club fitness routine. He experienced a progressive loss of hip mobility.MRI scanning and radiographs of the left hip joint were compatible with Kellgren-Lawrence Grade 3 osteoarthritic changes of the left hip joint. On physical examination, he had an antalgic limp and a positive Trendelenburg sign. Passive ROM of the hip joint was associated with the reproduction of severe groin pain, crepitus, and a loss of internal rotation. The patient had a BMI of 27. NSAIDs had failed to provide adequate pain relief. The patient was seriously considering total hip arthroplasty. In an attempt to avoid surgery, he elected to have an injection of an EVIP containing active GFs and exosomes into his hip.
Read the full article at here.
Mitchell B Sheinkop and Maxwell Dordevic.2019, Intra-Articular Injection of An Extracellular Vesicle Isolate Product to Treat Hip Osteoarthritis. Int J Recent Sci Res. 10(12), pp. 36230-36232. DOI: http://dx.doi.org/10.24327/ijrsr.2019.1012.4884
Tags: clinical trial, Osteoarthritis, patient results, published article

Feb 14, 2019
Following injury or wear, limitations need not be limited to professional, college and high school athletes
Last week, my column focused on the post traumatic ravages of the National Football League season; and getting ready for next cycle of body demolition. Those who watched the championship game saw several players assisted off the field following violent trauma. Even the President announced that he would prefer his son not play football. While professional, college and high school competitive sports enjoy high profile, there are the average recreational sports and fitness enthusiasts who progressively experience a diminution of ability to participate in a sought-after activity by virtue of injury or wear and tear; be it basketball, running, cycling, skiing, hockey, fitness endeavors, volleyball, golf, soccer, sailing; so, on and so forth.
As an example, a 67-year-old man presented in my office last week after having read my column, with progressive pain in his left knee and inability to partake in his long-time recreational passion, volleyball. I completed his intake centered on the taking of his medical history, completed an orthopedic physical examination, and ordered X-rays. He brought a recent MRI study to the appointment. The physical therapist who assists me objectively documented his physical findings so we would have pretreatment measurements. Additionally, my therapeutic recommendation is always based on more than an X-Ray and MRI, but also includes the objective and reproducible Range of Joint Motion and assessment of joint stability.
After review of all the above, I administered informed consent for a Bone Marrow Concentrate intervention into his left knee as a means of postponing, perhaps avoiding a Total Knee Replacement and assisting in his return to recreational volleyball. While there are several options for intervention into the arthritic or injured joint including adipose and blood-based alternatives, I recommended Bone Marrow Concentrate for his moderately arthritic knee as a means of providing Concentrated Platelets, Concentrated Growth Factors, anti-inflammatory Cytokines, Adult Mesenchymal Stem Cells, Precursor Cells, Marrow Adipose Tissue, and Hematopoietic Cells, all consistent with FDA compliance requirements. There are mandatory Federal Drug Administration and Federal Trade Commission regulatory standards of compliance in Regenerative Medicine; patients must be cautious when choosing the minimally invasive Cellular Orthopedic option. It takes an office visit, physical assessment and review of images by a board-certified specialist in the regenerative medicine decision making process; not merely attendance at a seminar. Some patients will not meet inclusion criteria, their needs would be better served by accepted surgical norms.
At age 67, there is little chance at Cartilage Regeneration for my patient, but there is a high probability of Joint Restoration; that is increased motion, diminution of pain and the return of ability, in this patient’s case to play volleyball. He will wear an off-loader brace for six weeks, partake in physical therapy and then gradually return to his Chicago Park District three games a week routine. The patient will have reached his competitive goals through care based on the scientific evidence and outcomes documentation. As long as I introduced the subject of competitive goals, off I go to ski in Colorado next week; about a year after having undergone Cellular Orthopedic interventions to both of my arthritic hips and both of my arthritic knees. Without the biologics, not only would I be unable to ski, I would be ready for two knee replacements and two hip replacements. I should have listened to my mother and stuck to the piano and violin.
To learn more about continuing to reach your competitive goals, visit my web site www.sheinkopmd.com or call and schedule a consultation 312-475-1893).
Tags: arthritis, board-certified, Bone Marrow Concentrate, bone marrow edema, cellular orthopedics, FDA, football, Growth Factors, hematopoietic cell, joint pain, Mesenchymal Stem Cell, OA, Orthopedic Surgeon, Osteoarthritis, Physical Therapy, soccer, sports injuries, sports medicine, tear, volleyball
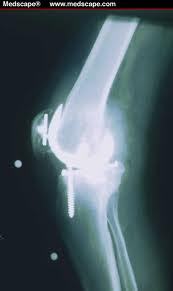
Oct 5, 2018
After reviewing a CT scan of the right knee where a Total Knee Replacement had been done provided by a patient who sought my opinion for stem cell treatment of her arthritic left knee, I called her to discuss the possible reasons that the right Total Knee Replacement that she had undergone over ten years ago was never satisfactory. At the time of the right TKR for arthritis, she had elected for what seemed at the time as a logical intervention for a painful, arthritic, right knee. The outcome was at first complicated by a lot of postoperative swelling and pain; at one-year post-operative, the patient had never been pain-free nor had she ever regained her preoperative range of motion and that adverse outcome has persisted for over ten years. Fast forward a decade and the right knee has continued to be a source of pain and limited motion; so much so that the patient decided to seek consultation for a Stem Cell treatment for her now arthritic left knee.
Her first question was basically “does stem cell therapy for knees work?”. Our data collected on patients treated in this office over the past six years, since I started my practice, is compatible with an 85% patient satisfaction rate. We have not recorded one complication. To be forthright, not every patient has a perfect result; but the vast majority experience marked reduction in pain, increased motion and a significant improvement in functional capacity. Unlike a failed outcome of a Total Knee Replacement, our stem cell injections may be repeated at anytime if the benefits of the initial intervention do not last.
When it comes to the cost of a stem cell intervention, while the Total Joint Replacement is more often than not covered by some type of insurance, our fee schedule is in keeping in line with the deductible a patient has to pay out of pocket for the major surgery. If you do the math and additionally compare the risks and benefits of a stem cell intervention with a total knee replacement, you will note:
- Remote to little risk of complication with a stem cell procedure
- Comparable cost comparing my charges for a stem cell intervention with the inherent deductible obligation for a Total Joint Replacement
- Rapid recovery with a stem cell intervention versus the risks of an adverse outcome as experienced by the patient seeking consultation for stem cells
To seek consultation call (312) 475-1893. You may visit my website at WWW.sheinkopmd.com
Tags: cellular orthopedics, ct scan, joint health, joint replacement, knee pain, knee replacement, Osteoarthritis, regenerative medicine, stem cell treatment, TKA
Jun 22, 2018
My regenerative and restorative Cellular Orthopedics practice is for the most part, evidence based. By that I mean, the outcomes data collected over these past five years regarding the several thousand patients with skeletomuscular afflictions that I have treated with a selection of alternatives using a needle and not a knife is generally based on regenerative and restorative interventions. While not everyone has experienced a dramatic change in symptom relief and functional improvement, many have. The statistical outcomes evidence follows a bell shaped curve with some experiencing immediate improvement as I have in both my hips and knees, while most take several weeks or longer with a continuing improvement up to 18 months post intervention. While it is true that five percent of patients are not satisfied after several years and have gone on to a joint replacement, 95% of my patients are well satisfied and have returned to, or never quit doing what they love.
At the onset of my cellular orthopedic initiative, the interventions were solely based on Platelet Rich Plasma options and Bone Marrow Concentrate; today, our menu of services can be customized so as to meet the needs of all seeking to improve the quality of life and avoid a major surgical procedure. Not only can I concentrate PRP as needed, I can customize the concentration to meet a patient’s particular needs using hemo-analysis. Bone Marrow Concentrate rich in Adult Mesenchymal Stem cells, Platelets, Growth Factors and Precursor Cells is still the foundation of my practice, however for the past year, I am able to offer a Platelet Concentrate derived Growth Factor and Protein Solution option when indicated.
Then there are those whose co-morbidity or prescription medication dependency excludes them from the aforementioned autologous choice of options. As of this upcoming Tuesday, I have acquired an intervention technology that will help patients seeking to a void a total joint replacement who are not candidates for existing regenerative medial offerings. There are many reasons to explain a 5% failure rate including genetic cartilaginous variations, any bell shaped curve will have a small number who don’t pass the final examination. Incidentally, if and when such occurs, I offer another intervention frequently at no charge or certainly at a discounted rate.
Should you want to learn more or schedule a consultation, call
(312) 475-1896. You may visit my web site where you will find the webinar at www.Ilcellulartherapy.com.
Tags: bone marrow, cellular orthopedics, Growth Factors, Hip pain, joint pain, joint replacement, knee pain, Mesenchymal Stem Cell, Osteoarthritis, platelets




