Jan 8, 2021
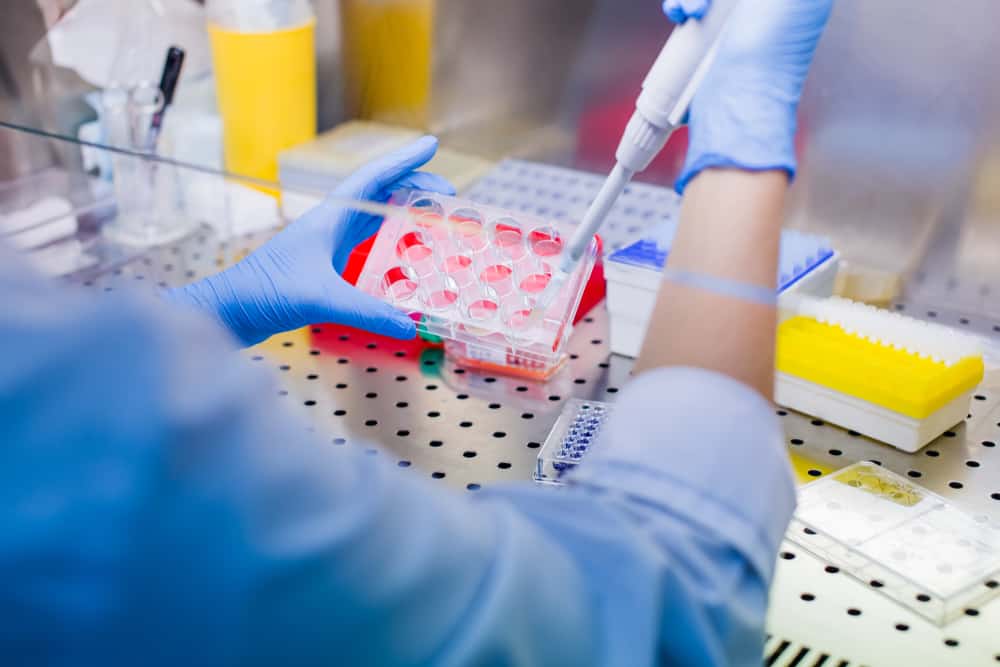
“I am sitting here at the office, just finishing the first draft of the clinical study report for PSC-CP-001. I am deeply moved and grateful for the incredible efforts you have all made in advising, enrolling patients, completing data forms, monitoring, and all other manner of work to bring this glimmer of an idea from 2018 to a completed first study in 2020. We overcame the FDA, the reluctance of patients to enroll, and the personal and professional perils of COVID-19 pandemic. Many studies and companies were just shuddered. You all kept the faith and pivoted to Zoom and remote exams, along with the help from FDA to allow us to keep moving. In the midst of all this, we also managed to file a new IND for COVID-19, manufacture the cells, get FDA approval, and start the study (2 patients already treated and high hopes). Honestly, Chris and Sue have been the steady Eddy’s for me assuring you all had the right resources and instructions, and each of the site coordinators are Saints. And Carolyn’s manufacturing crew work OT to make sure we extracted the most cells and had a safe product to ship. And Mike has raised capital and kept the company running including doing an amazing license deal that brought us additional capital to keep burning the midnight oil.
The data is awesome. The cells were safe. Not a single Serious Adverse Event. Great data. In the coming week or two I will shepherd the final report through data analysis, editing, investigator review (yes you get to review!) and final QC/QA review. With final signatures it will go to FDA. Shortly behind this submission will be the request to conduct a full blinded placebo-controlled and randomized trial.”
Bob Harman, DVM, MPVM
CEO, Personalized Stem Cells, Inc.
CEO, VetStem Biopharma, Inc.
Personalized Stem Cell Clinical Trial Updates
In 2021, we also plan quickly to apply for IND (FDA abbreviation for Investigational Device Exemption) for cultured adipose cells for knee arthritis. Later in the year, we plan to apply for Clinical Trials to treat multiple orthopedic conditions including knees, hips and shoulders. More specific information will be posted on this Blog as applications are submitted and/or approved. In the meantime, while you are waiting, our Stem Cell, Platelet Rich Plasma and Growth Factor options are available to relieve arthritic symptoms and improve your functional capacity. While I am waiting for my participation in several of the upcoming trials for hips and knees and hopefully spine, either this week or next, I will be undergoing a proprietary Platelet Rich Plasma /Growth Factor intervention at the knee to enable me to ski in February without restrictions.
To learn more about my treatment options, visit my web site at www.Sheinkopmd.com.
To schedule an office visit, call (312) 475-1893. To stay updated on the Clinical Trial developments, don’t miss reading my weekly Blog
Tags: clinical trial update, clinical trials, stem cell clinical trials, stem cell therapy

Jun 22, 2020
“They are close to reaching the max number in the study and feel they have enough data at this point.”
While we still could offer adipose derived stem cells to patients via Liposuction and processing of the adipose tissue to recover millions of stem cells, the procedure will revert to fee for service. In the post PSC Trial treatment protocol, it would be possible to address several joints in the same setting with adipose derived cells. Owing to the changes in the PSC Trial protocol necessitated by the COVID-19 virus mandated quarantines, the FDA has diminished the number of patients needed to be enrolled. While I have not yet harvested the adipose tissue from all current enrollees, nor have I injected the stem cells in those already having gone liposuction, we will be done harvesting and delivering the stem cells by mid-summer and have done our part.
Personalized Stem Cells Trial Update
At the beginning of the Trial last August, PSC became the first FDA approved clinical trial in the United States allowing the seven investigative sites to create a drug from a patient’s tissue for the treatment of arthritis. As it stands now, all biologics will be reclassified as drugs by the FDA as of November, 2020 and Personalized Stem Cells became the leader in this endeavor. It will not be for another six months that our team of seven clinical trial sites will have the necessary outcomes data required which would allow the FDA to reclassify the stem cells recovered from your adipose tissue as an approved drug.
On another note, yesterday, Northwestern Memorial Hospitals announced a “successful double lung transplant” to treat a young patient whose lungs were destroyed by the the COVID virus. In a previous Blog, I had introduced the PSC initiative to treat COVID pneumonia with adipose derived stem cells. That trial is underway in San Diego. One of the advantages enjoyed by those who are enrolled in the knee arthritis trial is storage of their stem cells. Those stored cells are available to enrollees for the future care of an arthritic joint in addition the study knee; but as well, for systemic disease outside of the musculoskeletal system such as COVID pneumonia. There are stem cells harvesting and storage companies cropping up on the internet; but as of this time, only Personalized Stem Cell, Inc.is allowed to treat. Should this pandemic continue, a patient at risk, be it age related or health related, for COVID complications just might contact our office and arrange for a fat graft harvest and storage of those adipose derived stem cells to meet a future need.
To schedule a consultation for the care and treatment of your symptomatic joint with limited function, call (312) 475-1893. You may visit my web site and watch my Blog at www.sheinkopmd.com To learn more about Personalized Stem Cells and the COVID lung trial, visit PersonalizedStemCells.com
Tags: clinical trials, personalized stem cells, stem cell clinical trial

Jan 13, 2020
DEFENSE HEALTH AGENCY 7700 ARLINGTON BOULEVARD, SUITE 5101 FALLS CHURCH, VIRGINIA 22042-5101 MEMORANDUM FOR DEPUTY CHIEF, READINESS AND HEALTH, BUREAU OF MEDICINE AND SURGERY DEPUTY CHIEF OF STAFF G-3/5/7, OFFICE OF THE SURGEON GENERAL DIRECTOR, MEDICAL OPERATIONS AND RESEARCH, OFFICE OF THE SURGEON GENERAL DIRECTOR, NATIONAL CAPITAL REGION MEDICAL
DIRECTORATE SUBJECT: Use of Regenerative Medicine Advanced Therapy (RMAT) Products Not Approved by the Food and Drug Administration (FDA).
Regenerative Medicine Advanced Therapy (RMAT) products not approved by the FDA have the potential to expose patients to unnecessary risk. One such product is injectable micronized dehydrated human amnion/chorion membrane (dHACM) used to treat conditions such as plantar fasciitis, tendonitis or osteoarthritis. Any of these products not approved by the FDA are considered investigational, at present. Therefore, effective immediately, all MHS facilities will stop using injectable RMAT products not approved by the FDA, including dHACM. Once the FDA has approved an RMAT product, further review will occur. The Veteran’s Administration has taken a similar action.
PAUL R. CORDTS, MD Deputy Assistant Director – Medical Affairs Defense Health Agency
At the same time I was made aware of the above Defense Department Memorandum, a patient called my attention to a brochure she had received in the mail on the same day recruiting “victims” to one of several offices in metropolitan Chicago treating patients with the very product that the Defense Health Agency classified as unapproved by the FDA and therefore prohibited for safe use. A day later. I received an e-mail from a health care provider out of state, a non-MD asking me about use of “stem cells and amniotic fluid in my office.”
Are these alleged members of the health care community charlatans, uninformed, or just plain dumb and dumber? “Beware, take care, of the Green-eyed dragon with 13 tails, he’ll feed, with greed”. There is only one FDA approved source of stem cells for arthritis available in the United States and I am pleased to have been designated a Clinical Investigator in the PSC Trial. You may read about it on my website at www.sheinkopmd.com or at www.personalizedstemcells.com . The other potential biologic alternatives that, to the best of my knowledge are approved at this time, involves using your bone marrow as a regenerative option for delivering Adult Mesenchymal Stem Cells, or Platelet Rich Plasma obtained from your circulating blood as resource for Growth Factors.
To learn more or to schedule a consultation, call (312) 475-1893
Tags: clinical trials, FDA approved stem cell research, regenerative medicine, stem cell research

Jan 3, 2020
The complexities inherent in biologics, cellular therapies, and stem cells as therapeutic entities are being been addressed at a number of levels. Thoughtful editorials, consensus statements, and even actions by state licensing boards, the FDA and the FTC for unprofessional conduct are starting to “drain the swamp.”
A large part of this growing industry consists of off-label uses; many in Regenerative Medicine seem to practice without FDA compliance. There are some indications where biologics and cellular therapies show genuine promise such as osteoarthritis, and my ongoing clinical research is conformational. Still owing to the bad actors, charlatans, and camp followers, substantive questions remain, including some basic concerns about safety, efficacy, cell sourcing and delivery, host response, and mechanism of action. There is only one way to approach this problem, and that is research with FDA governance (Personalizedstemcells.com).
In general, people thrive on novelty. We look for the latest and newest forms of treatment, hoping that the just-released remedy with the special formula will be the one to cure our problems and ease our pains. We do this as patients, and we do it as physicians. My major concern is that anyone licensed by the Department of Education and Registration may attend a weekend course, upload a web site, and begin fleecing an unsuspecting public.
Musculoskeletal disease and the resulting pain and loss of function are an invitation to the charlatans and camp followers: 20% of the population experiences arthritis and other serious musculoskeletal diseases. it’s not surprising that patients (and providers) will look anywhere for relief, and the bad actors will take advantage of an opportunity.
In the past, this approach has not worked well for many patients. The list of failed treatments for common musculoskeletal diseases is a long and sad one, from nutraceuticals to chelation therapy, and diets from alfalfa to zinc. At times, orthopaedic surgeons have not been careful stewards of clinical evidence—we continue to use viscosupplementation, despite the paucity of good evidence and we continue to use arthroscopic knee surgery in patients who should not have it. It would be nice, after all this, to get it right vis-à-vis stem cells and related treatments.
It appears that government sanctions, editorial sermons, and statements from specialty groups have not arrested the many dubious practice patterns we’ve all seen and too many patients experience. Witness the Amniotic Fluid debacle in Illinois last year perpetrated by the Bad Actors wherein patients were paying astonishing amounts of money (and taking yet-unquantified risk) for treatments that were of no benefit. The Amniotic Fluid marketed at seminars had and has no viable stem cells or biologics and cell therapies for regeneration of musculoskeletal tissues. The same holds true for Cord Blood and Wharton’s Jelly as evidenced by the epidemic of infections experienced in 2018 and 2019.
In this new decade, be informed, protect yourself, and seek out Board Certified Specialists in Regenerative Medicine if you want to postpone, perhaps avoid a joint replacement; or if you want to maintain or return to a fully active lifestyle. To schedule an appointment, call (312) 4745-1893. You may learn more at www.sheinkopmd.com.
Tags: biologics, cellular therapy, clinical trials, opinion on stem cell threeapy, stem cell therapy



