Oct 5, 2018
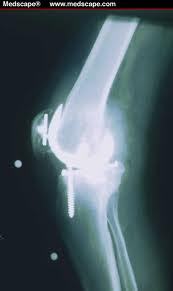
After reviewing a CT scan of the right knee where a Total Knee Replacement had been done provided by a patient who sought my opinion for stem cell treatment of her arthritic left knee, I called her to discuss the possible reasons that the right Total Knee Replacement that she had undergone over ten years ago was never satisfactory. At the time of the right TKR for arthritis, she had elected for what seemed at the time as a logical intervention for a painful, arthritic, right knee. The outcome was at first complicated by a lot of postoperative swelling and pain; at one-year post-operative, the patient had never been pain-free nor had she ever regained her preoperative range of motion and that adverse outcome has persisted for over ten years. Fast forward a decade and the right knee has continued to be a source of pain and limited motion; so much so that the patient decided to seek consultation for a Stem Cell treatment for her now arthritic left knee.
Her first question was basically “does stem cell therapy for knees work?”. Our data collected on patients treated in this office over the past six years, since I started my practice, is compatible with an 85% patient satisfaction rate. We have not recorded one complication. To be forthright, not every patient has a perfect result; but the vast majority experience marked reduction in pain, increased motion and a significant improvement in functional capacity. Unlike a failed outcome of a Total Knee Replacement, our stem cell injections may be repeated at anytime if the benefits of the initial intervention do not last.
When it comes to the cost of a stem cell intervention, while the Total Joint Replacement is more often than not covered by some type of insurance, our fee schedule is in keeping in line with the deductible a patient has to pay out of pocket for the major surgery. If you do the math and additionally compare the risks and benefits of a stem cell intervention with a total knee replacement, you will note:
- Remote to little risk of complication with a stem cell procedure
- Comparable cost comparing my charges for a stem cell intervention with the inherent deductible obligation for a Total Joint Replacement
- Rapid recovery with a stem cell intervention versus the risks of an adverse outcome as experienced by the patient seeking consultation for stem cells
To seek consultation call (312) 475-1893. You may visit my website at WWW.sheinkopmd.com
Tags: cellular orthopedics, ct scan, joint health, joint replacement, knee pain, knee replacement, Osteoarthritis, regenerative medicine, stem cell treatment, TKA
Dec 20, 2017
Since we practice in an emerging discipline of Regenerative Medicine, how is regeneration determined? Cartilage repair should be evaluated with use of a scoring system that considers the volume of the defect that becomes filled with repair tissue, the integration of repair tissue with adjacent cartilage, and the macroscopic appearance and biomechanical properties of the repair site. The macroscopic assessment is particularly important in evaluating cartilage repair because it provides information about the quality of the full repair site compared to the incidental histological assessment which only evaluates a biopsy of the repair site.
If the aforementioned answer to my introductory question may seem scientifically oriented, that is purposeful on my part; because only those able to explain Regenerative Medicine on a clinical, technical and scientific basis should be caring for your arthritic joint.
While an arthroscopic evaluation provides the best opportunity for a determination of joint regeneration 18 months or greater following a Bone Marrow or Platelet Rich Plasma or other cellular orthopedic intervention for arthritis, an invasive surgical evaluation is not realistic. For a quantitative MRI to assist in the assessment requires availability of a specialized imaging center and there are just too many variables to allow for dependable quantitation; expense is prohibitive. The most dependable and reproduceable means of measuring the arthritic or injured joint response to a cellular orthopedic intervention is a history and physical examination, the latter completed with a tape measure and goniometer as well as an activity assessment. By comparing a baseline measurement prior to an intervention and at serial intervals following the procedure, one may determine if regeneration is indeed taking place and thus establish clinical practice guidelines and determine Evidence Based Quality and Value.
Now for the real question, does cartilage regeneration need to take place on a macroscopic level for cellular orthopedics to succeed? New therapies such as bone marrow derived stem cells, growth factors and cytokines; platelet-rich plasma (PRP); and IRAP (interleukin-1 receptor antagonist protein) first and foremost address the bio-immune basis of degenerative arthritis. By controlling the pain and eliminating inflammation; stopping the progression (at least slowing) of Osteoarthritis; reversing scarring, thus improving motion and function; and lastly, possibly regenerating cartilage for those in whom regeneration is possible. From the editors of the Encyclopedia Britannica: “Interleukin (IL), any of a group of naturally occurring proteins that mediate communication between cells. Interleukins regulate cell growth, differentiation, and motility. They are particularly important in stimulating immune responses, such as inflammation.”
Should our future blogs and discussions address not cartilage regeneration but rather reversing the proinflammatory cytokine production from the synovial lining of the inflamed knee? One such possible pharmacological treatment of OA is anticytokine therapy. Interleukin-1 (IL-1), as a main inflammatory and catabolic cytokine in the pathophysiology of OA, represents one of the possible treatment targets. Koby Bryant was one of the first highly visible professional athletes who travelled to Germany over eight years ago for Interleukin-1 Receptor Antagonist Protein intervention for an arthritic knee. Many, have followed including golfer Fred Couples for his problematic back.
If this Blog has introduced new considerations and questions, then let me clarify. Call 312 475 1893 to schedule an appointment. You may watch my webinar at www.Ilcellulartherapy.com
Tags: arthritis, bone marrow, Hip Replacement, Interleukin, Interleukin-1 Receptor Antagonist, IRAP, Knee Pain Relief, Osteoarthritis, stem cell treatment, stem cells, Subchondroplasty
Nov 29, 2017
I am being forthright; based on my review of data, while 80% or more of my patients continue to enjoy
satisfactory outcomes at four years or more following a stem cell intervention, there are those whose
symptoms and functional limitations recur. Please be aware that when I undertake the care and
treatment of a patient with a symptomatic and function limiting joint, it is with the notion of
regeneration and long-term benefit. It doesn’t always happen; there are may possible explanations.
Most important though is the need to identify possible causes of potential failure at the beginning, and
that is why we have recommendations before and after a procedure as to how to manage alcohol, diet,
supplements and a rehabilitation protocol. We also review your past medical history to identify any
possible indication that your stem cells have been adversely affected by co-morbidity or prior
treatments.
Assume if you will that you adhered to the initial pre-and post-intervention protocol but now returned
to my office months or years later with recurring symptoms. First and foremost is an updated medical
history and physical examination. That is followed by repeat images including X-rays and an MRI.
Mechanical progression of joint injury may result from aggravation of the preexisting damage by
subsequent trauma. Then there is the reality of identifying new processes within or adjacent to the joint.
This morning, I returned the phone call of a southwest Wisconsin dairy farmer; not the same patient I
wrote about last week. He has been a patient for over four years with a full restoration of work related
activities and recreational pursuits following several regenerative interventional options. After three
hours of basketball, three weeks ago, his knee pain returned. I called him back while he was milking his
cows and it was the first time I have been “mooed” at over a cell phone. I requested that the patient
update his X-rays, MRIs and then allow me to reevaluate him. A repeat stem cell intervention with a
more advanced technology, a subchondroplasty in addition to the stem cell intervention of his joint?
The recommendations will be based on an updated evaluation. In my practice of cellular orthopedics, it
isn’t one and done. Additionally, some of the more advanced techniques are being covered in part by
health care insurance
If you want to learn more, call for an appointment (312)475 1893
You may access my web site at www.Ilcellulartherapy.com and watch my webinar
After I completed writing this Blog, I opened the Bone and Joint Newsletter.
Lead article: Study Suggests Knee Replacement Be Reserved for Those More Severely Affected by Osteoarthritis. A recent analysis found that the current practice of TKR as performed in the USA had minimal effects on quality of life and quality adjusted life years
Tags: arthritis, Benefits and Risk, bone marrow, Bone Marrow Concentrate, Clinical Studies, Clinical Trial. Mitchell B. Sheinkop, Concentrated Stem Cell Plasma, Growth Factors, Hip Replacement, Interventional Orthopedics, Knee Pain Relief, Mesenchymal Stem Cell, Orthopedics, Osteoarthritis, Pain Management, Platelet Rich Plasma, regenerative medicine, stem cell treatment
Nov 22, 2017
As regular readers of this Blog are aware and as well, those patients who seek consultation in my office, my cellular orthopedics practice is based on clinical evidence. While the largest marketer of stem cells in the Midwest, bases a recommendation for a product without scientific evidence or even anecdote, my practice is evidence based. While it is true that I can site anecdote after anecdote about a successful outcome, the scientific process requires that I provide statistical support for what I do. The subject of this particular blog is to share with you how far I will go to gather that evidence.
About six months ago, I completed a combined Bone Marrow Concentrate Intra-articular (into the joint) /Subchondroplasty (into the bone) in the right knee of a dairy farmer, in his early 60s, living in Southwest Wisconsin. He was able to follow up at my office in Des Plaines twice following the intervention; but owing to the fall harvest, he had been unable to return for the third follow up in a timely manner. My desire is to assure the success of the procedure, rather than having completed a telephone interview, my wife and I headed out this past weekend to Dodgeville, Wisconsin, where I completed the full evaluation in a booth at the local Culvers. In addition to the usual historical review of progress, I completed a Physical Examination of the patient including circumferential measurement of the thigh, knee and calf using a tape measure and an assessment of his range of motion using a goniometer for accuracy. I think that the diners at Culvers were just as fascinated at what was taking place in our booth as I was watching the customers in the drive through ordering an early lunch while carrying a recently harvested buck in the back of their pickups. It is deer season in Wisconsin.
Suffice it to say that my patient was thrilled at my willingness to save him a trip to Chicago but he was even happier about the outcome in his arthritic knee. I was thrilled at his response to care. Whereas prior to the right knee intervention, he had constant pain and limitation in function, at six months, the pain and swelling are gone and he has unlimited function be it on stairs, kneeling or climbing in and out of the tractor. What separates me from the madding crowd is the data and evidence for which my recommendations are advanced. Incidentally, two blogs ago, I listed our clinical trials, the basis for our evidence based practice. Below is the trial in which my Wisconsin patient is enrolled and for which I sought follow up:
- Stem Cell Counts and the Outcome of Bone Marrow Concentrate intra-articular and intra-osseous (subchondroplasty) interventions at the knee for grades 2 and 3 OA. (supported in part by Celling). Ongoing
His results are so good that his wife decided to be next in line for her knee that did not respond well to arthroscopic surgery two years ago.
Whether you didn’t respond to surgery or haven’t had surgery, call 312 475 1893 to schedule an appointment or watch my webinar on the website www.ilcellulartherapy.com
This Thanksgiving week, let me give thanks to those who read this Blog
Tags: arthritis, bone marrow, Bone Marrow Concentrate, cellular orthopedics, Clinical Studies, Clinical Trial. Mitchell B. Sheinkop, Knee Pain Relief, Mesenchymal Stem Cell, Osteoarthritis, regenerative medicine, stem cell treatment

Nov 9, 2017
The other day, I noted a www.Ilcellulartherapy.com (my web site) request from a patient seeking to schedule an appointment at my office. That phone call had been placed ten days earlier and my assistants failed to respond in a timely manner. When eventually returned, the potential patient indicated that she had decided to seek consultation elsewhere. I can understand her frustration and yet would her orthopedic needs have been better served by placing a second request? I share this with you and admit we are human; as hard as we try in achieving 100% patient satisfaction, at times the system doesn’t operate as intended. The ultimate question is where to seek Cellular Orthopedic consultation and treatment even if initially, the response is not as either side would like?
For almost five years of a Cellular Orthopedic, Regenerative Medicine practice, I have integrated patient care with clinical research. This is my contribution to the evolving discipline of Stem Cell therapy for arthritis; and is based on what I introduced into orthopedic surgery during my 37-year academic joint replacement career. It is only by monitoring outcomes that we continue to learn and improve results.
Listed below are my clinical research endeavors over the past five years and the basis for my clinical recommendations:
1) Stem Cell Counts and the Outcome of Bone Marrow Concentrate intra-articular and intra-osseous (subchondroplasty) interventions at the knee for grades 2 and 3 OA. (supported in part by Celling). Ongoing
2) Outcomes of Bone Marrow Concentrate (stem cell, platelet and growth factor) Intervention at the Knee for Grades 2 and 3 OA in 50 patients at 2 to 4 years. (supported in part by Regenexx)
3) Outcomes of Intra-articular Bone Marrow Concentrate versus those of combined Intraarticular and Intraosseous interventions for grades 2 and 3 OA at the knee at one year. (self-funded). Ongoing
4) How does the PRP and Mononucleated cell count affect the outcome of a BMC intervention for grades 2 and 3 Knee OA? (a joint project with Greyledge) Ongoing
5) Safety and Efficacy of Percutaneous Injection of Micro-Fractured Adipose Tissue for grade 4 Osteoarthritic Knees, minimum follow-up of 18 months in 30 patients (supported in part by Lipogems)
6) A Lyophilized Amniotic Fluid Clinical Trial for Grades 2 and 3 Knee OA (supported by MiMedx) Ongoing
7) An Amniotic Fluid Cryopreserved Clinical Trial for Grades 2 and 3 Knee OA in 30 patients. (supported by MiMedx)
8) An Amniotic Fluid Cryopreserved Clinical Trial for Grades 2 and 3 Knee OA in 45 patients. (sponsored by Liventa)
We Have The Evidences for Cellular Orthopedics; ask the camp followers to show you theirs.
To schedule an office visit call 312 475 1893 and please be patient.
Tags: cellular orthopedics, Clinical Trial. Mitchell B. Sheinkop, regenerative medicine, stem cell treatment, stem cells, Subchondroplasty

