Feb 21, 2019
We read about it in the sports pages every day; the player presenting after twisting a knee, feeling a “pop” and going down in a heap. The knee is swollen, the first indication that bleeding has occurred inside the joint. Physical examination of the knee to test the ligaments leads to a suspected tear of the anterior cruciate ligament; the MRI is ordered and confirms the diagnosis.
Because of its poor blood supply and location inside the knee, the ACL has little healing potential. It’s an unfortunate reality, as they are occurring at increasing rates over the past two decades. In part, it’s because more children are playing competitive sports and doing so at a younger age while we seniors are still skiing or playing and competing all year. What are the future implications of a torn ACL? Lindsey Vonn came back after ACL surgery; so did Tiger Woods, Julian Edelman, Tom Brady and Derrick Rose to name a few. so how bad can it be? The truth is that surgery can restore knee function, but it does little to diminish the risk of arthritis 10 to 15 years down the line or less. Lindsey Vonn announced retirement five years later because of arthritis. Kids who tear their ACL today are often left with 60-year-old knees when they’re 30; and as has been recently stated, Knee Replacement is not necessarily a panacea.
Secondary damage may occur in patients who have repeated episodes of instability due to ACL injury. With chronic instability, a large majority of patients will have meniscus damage when reassessed 10 or more years after the initial injury. Similarly, the prevalence of articular cartilage lesions increases in patients who have a 10-year-old ACL deficiency. It is common to see ACL injuries combined with damage to the menisci, articular cartilage, collateral ligaments, joint capsule, or a combination of the above; the “unhappy triad,” especially in football players, soccer players, basketball players and skiers.
Certainly, modern ACL surgery means it’s no longer a career-ender, but recovery ranges still vary widely. In cases of combined injuries, surgical treatment is warranted and generally produces better outcomes. As many as half of meniscus tears may be repairable and may heal better if the repair is done in combination with the ACL reconstruction. Some athletes come back in as little as nine months, while it can take well more than a year for others. Then comes the mental battle, that is the silent war waged after tearing an ACL. Derrick Rose, we are watching you.
Recent clinical evidence suggests surgery is not your only option; interventional or cellular orthopedics may be a non-surgical alternative that uses your own cells to repair the incompletely damaged ligament. A cellular orthopedic intervention for those who meet the inclusion criteria may substitute for surgery. Those who offer the non-operative option when appropriate, use Bone Marrow or Adipose Tissue harvested from your skeleton or abdomen, process the recovered cells and growth factors with particular attention to FDA compliance, and inject the concentrate into the remaining Anterior Cruciate Ligament cumented incomplete tears with success in returning athletes to a sport.
While intervention and cellular orthopedics may have a role in a torn ACL at times, Joint Restoration, perhaps even Regeneration adjuncts at the time of an ACL repair or for the ensuing arthritis have an absolute evidence-based role. To schedule an appointment call (312) 475-1893. You may visit my web site at www.sheinkopmd.com
Tags: ACL, ACL Injury, anterior cruciate, arthritic knee, cellular orthopedic
Jul 19, 2018
The argument frequently advanced by orthopedic surgeons in response to a patient’s inquiry concerning stem cells for arthritis is that it is too early, there is not enough research, It is better to have a major surgical procedure. For those of you who have read my blog or have sought orthopedic consultation in my office, I have emphasized that my recommendations are evidence based. Each patient, for whom I have completed a cellular orthopedic intervention for arthritis, has been entered into a registry or clinical outcomes data base, IRB approved. Just as I pioneered the integration of clinical care with clinical research over 37 years as a joint replacement surgeon, so too do I now partake in the growth and development of the clinical pathways for regenerative medicine.
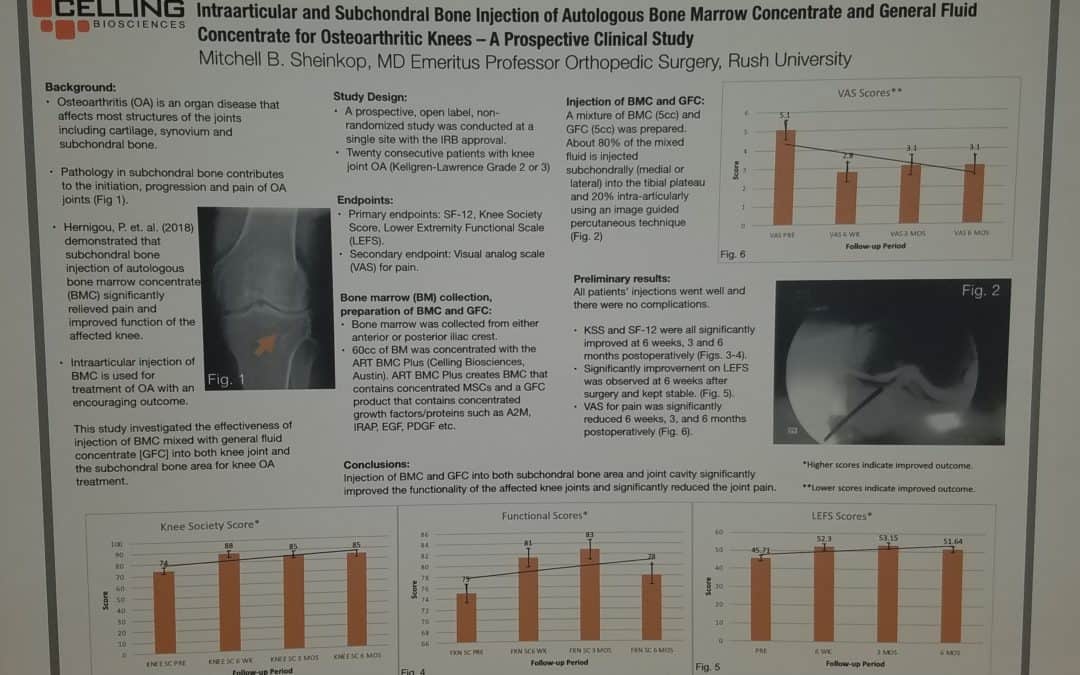
Last month, I exhibited a poster at a large regenerative medicine meeting wherein I shared my preliminary outcomes and thus educated other professionals using Intraarticular and Subchondral Bone Injection of Autologous Bone Marrow Concentrate and General Fluid Concentrate for Osteoarthritic Knees-A Prospective Clinical Study. Osteoarthritis is an organ disease that affects most structures of joints including cartilage, synovium and subchondral bone. Pathology in subchondral bone contributes to the initiation, progression and pain of Osteoarthritis. In previous European studies, the injection of autologous bone marrow concentrates into bone supporting the joint significantly relieved pain and improved function of the affected knee. The preliminary outcomes in the study that I presented via a poster exhibit, investigated the effectiveness of injections of Bone Marrow Concentrate with General Fluid Concentrate (Growth factors), into both the knee joint and the subchondral bone. The study recorded all the standard Endpoints I had previously used in joint replacement clinical outcomes trials.
Bone Marrow was collected from the pelvis and a filtration system allowed for concentration of Mesenchymal Stem Cells, Platelets, Precursor Cells and Growth factors such as A2M, IRAP, EGF, PDGF, TNF-B blocker, etc. After preparation, a mixture of Bone Marrow Concentrate and Growth factor Concentrate was injected into the bone (subchondral) and into the joint.
In the study, all patient injections went well and there were no complications. The Preliminary Results documented diminished pain and improved function. We concluded that injection of Bone Marrow Concentrate and Growth factor Concentrate into both the subchondral bone area and joint cavity significantly improved function of the affected knee joints and significantly reduced joint pain. While there are many stem cell providers to be found because of their marketing, choose the center of excellence in Cellular Orthopedics that is evidence based.
Call to schedule a scientific based consultation from an orthopedic surgeon 1 (312) 475-1893.
You may access my web site at www.SheinkopMD.com.
Tags: avascular necrosis, bone lession, bone marrow, Cartilage, cellular orthopedics, clinical study, Growth Factors, IRAP, joint pain, joint replacement, knee pain, knee replacement, meniscus tear, Osteoarthritis, platelets, PRP, regenerative medicine, sports medicine, stem cells, subchondral bone
Apr 5, 2018
If you remember those children’s song lyrics, you will march right up the skeleton. The orthopedic message is that what’s happening in your foot and ankle will affect the well-being of your knee and hip. I was reminded of the continuum on Tuesday when a patient I had treated in November of 2017 returned for follow up this past Tuesday. Once a prominent running back at the college level, he had presented 20 years after a “high ankle sprain” with a Talar Dome Lesion at the right ankle and early onset post traumatic arthritis; in plain speak, an injury to the cartilage and underlying bone. Not only did the right ankle impairment affect his foot and ankle, he was experiencing progressive pain in his knee and hip thus altering his gait, his fitness pursuits and forcing change in his recreational profile. Running was no longer possible nor was snowboarding.
Increasingly, these Talar Dome lesions or osteochondral injuries are being diagnosed long after what was thought to have been a sprained ankle. In the case of my patient, last November, I performed a minimally invasive procedure wherein bone marrow was aspirated from his pelvis, concentrated, processed, and injected into both the ankle joint and bone marrow defect of the talus under fluoroscopic guidance. Osteochondral injuries and bone marrow lesions are a continuum of small posttraumatic defects that pathologists have shown represent a failed healing response. Most readily diagnosed on an MRI, with time, a rim of sclerosis may develop so the abnormality may lend itself to diagnosis with an X-ray. This type of defect is not limited to the ankle and may be found throughout the extremities and pelvis. They may be found in any joint region that sees weight bearing or repetitive stress though; most commonly, they are associated with trauma as was the case, though long removed in my patient.
In the case of this vignette, on Tuesday I had determined that ankle and subtalar joint motions had become symmetrical. He no longer complained of pain; equally important, the bony defect and joint changes could no longer be seen on X-ray. In short, he had healed. In the past six months, I have followed two other equally rewarding Bone Marrow (stem cell/growth factor/platelet) intervention outcomes at the talus and more than six around the knee.
If you are experiencing joint pain and altered function without an explanation or in spite of a course of “conservative” treatment, it may be time to learn more about how Bone Marrow Concentrate, that is stem cells, platelets and growth factors, may relieve bone and joint pain, restore function and help you postpone, perhaps even avoid a major surgical procedure.
Call 312 475 1896 to schedule a consultation or visit my web site and watch the webinar at www.ilcellulartherapy.com
Tags: ankle pain, bone lesion, bone marrow, Cartilage, joint pain, Mesenchymal Stem Cell, Osteoarthritis, osteochondral defect, osteochondral injurie, Osteochondritis Dissecans, Subchondroplasty, Talar Dome lesions

Feb 16, 2018
I am sitting at my computer this morning writing the weekly Blog posting and not attending the IOF meeting taking place today in Broomfield, Colorado; yet I am reporting about the meeting. Instead of attending, I am preparing for a week-long ski adventure with my family next week in Vail, Colorado while trying to catch up in my practice. How is it than possible that I know what is taking place at the meeting? Listed below are five of the 10 ongoing or completed cellular orthopedic clinical trials in which I am a principal investigator or co-researcher. The preliminary and final data resulting from these clinical research initiatives is the outcomes foundation for what is being presented at the IOF podium today and tomorrow.

1) Stem Cell Counts and the Outcome of Bone Marrow Concentrate intra-articular and intra-osseous (subchondroplasty) interventions at the knee for grades 2 and 3 OA. (supported in part by Celling). Ongoing
2) Outcomes of Bone Marrow Concentrate (stem cell, platelet and growth factor) Intervention at the Knee for Grades 2 and 3 OA in 50 patients at 2 to 4 years. (supported in part by Regenexx)
3) Outcomes of Intra-articular Bone Marrow Concentrate versus those of combined Intraarticular and Intraosseous interventions for grades 2 and 3 OA at the knee at one year. (self-funded). Ongoing
4) How does the PRP and Mononucleated cell count affect the outcome of a BMC intervention for grades 2 and 3 Knee OA? (a joint project with Greyledge) Ongoing
5) Safety and Efficacy of Percutaneous Injection of Micro-Fractured Adipose Tissue for grade 4 Osteoarthritic Knees, minimum follow-up of 18 months in 30 patients (supported in part by Lipogems)
I had to prioritize; and since most of the arthritis data being presented is all or in part mine, I already know the subject matter. By staying home, I also found the opportunity to browse “stem cell” websites as suggested by ads in today’s newspapers or introduced by email blasts this week. Wow, a patient acting more like a consumer is really at risk for succumbing to Regenerative Medicine “false news”.
If you want to learn more about the difference between the stem cell purveyors and a legitimate, FDA compliant, evidence based, cellular orthopedics initiative, call to schedule a consultation or to get a second opinion.
You may schedule a visit at (312) 475-1893
You may access my website and watch a webinar at www.ilcellulartherapy.com
Tags: arthritis, bone marrow, Celling, cellular orthopedics, Growth Factors, Hip pain, International Orthopedics Foundation, joint pain, knee arthritis, knee intervention, knee pain, lipogems, Micro-Fractured Adipose, Osteoarthritis, PRP, regenerative medicine, Regenexx, stem cells, Subchondroplasty
Nov 29, 2017
I am being forthright; based on my review of data, while 80% or more of my patients continue to enjoy
satisfactory outcomes at four years or more following a stem cell intervention, there are those whose
symptoms and functional limitations recur. Please be aware that when I undertake the care and
treatment of a patient with a symptomatic and function limiting joint, it is with the notion of
regeneration and long-term benefit. It doesn’t always happen; there are may possible explanations.
Most important though is the need to identify possible causes of potential failure at the beginning, and
that is why we have recommendations before and after a procedure as to how to manage alcohol, diet,
supplements and a rehabilitation protocol. We also review your past medical history to identify any
possible indication that your stem cells have been adversely affected by co-morbidity or prior
treatments.
Assume if you will that you adhered to the initial pre-and post-intervention protocol but now returned
to my office months or years later with recurring symptoms. First and foremost is an updated medical
history and physical examination. That is followed by repeat images including X-rays and an MRI.
Mechanical progression of joint injury may result from aggravation of the preexisting damage by
subsequent trauma. Then there is the reality of identifying new processes within or adjacent to the joint.
This morning, I returned the phone call of a southwest Wisconsin dairy farmer; not the same patient I
wrote about last week. He has been a patient for over four years with a full restoration of work related
activities and recreational pursuits following several regenerative interventional options. After three
hours of basketball, three weeks ago, his knee pain returned. I called him back while he was milking his
cows and it was the first time I have been “mooed” at over a cell phone. I requested that the patient
update his X-rays, MRIs and then allow me to reevaluate him. A repeat stem cell intervention with a
more advanced technology, a subchondroplasty in addition to the stem cell intervention of his joint?
The recommendations will be based on an updated evaluation. In my practice of cellular orthopedics, it
isn’t one and done. Additionally, some of the more advanced techniques are being covered in part by
health care insurance
If you want to learn more, call for an appointment (312)475 1893
You may access my web site at www.Ilcellulartherapy.com and watch my webinar
After I completed writing this Blog, I opened the Bone and Joint Newsletter.
Lead article: Study Suggests Knee Replacement Be Reserved for Those More Severely Affected by Osteoarthritis. A recent analysis found that the current practice of TKR as performed in the USA had minimal effects on quality of life and quality adjusted life years
Tags: arthritis, Benefits and Risk, bone marrow, Bone Marrow Concentrate, Clinical Studies, Clinical Trial. Mitchell B. Sheinkop, Concentrated Stem Cell Plasma, Growth Factors, Hip Replacement, Interventional Orthopedics, Knee Pain Relief, Mesenchymal Stem Cell, Orthopedics, Osteoarthritis, Pain Management, Platelet Rich Plasma, regenerative medicine, stem cell treatment
Nov 22, 2017
As regular readers of this Blog are aware and as well, those patients who seek consultation in my office, my cellular orthopedics practice is based on clinical evidence. While the largest marketer of stem cells in the Midwest, bases a recommendation for a product without scientific evidence or even anecdote, my practice is evidence based. While it is true that I can site anecdote after anecdote about a successful outcome, the scientific process requires that I provide statistical support for what I do. The subject of this particular blog is to share with you how far I will go to gather that evidence.
About six months ago, I completed a combined Bone Marrow Concentrate Intra-articular (into the joint) /Subchondroplasty (into the bone) in the right knee of a dairy farmer, in his early 60s, living in Southwest Wisconsin. He was able to follow up at my office in Des Plaines twice following the intervention; but owing to the fall harvest, he had been unable to return for the third follow up in a timely manner. My desire is to assure the success of the procedure, rather than having completed a telephone interview, my wife and I headed out this past weekend to Dodgeville, Wisconsin, where I completed the full evaluation in a booth at the local Culvers. In addition to the usual historical review of progress, I completed a Physical Examination of the patient including circumferential measurement of the thigh, knee and calf using a tape measure and an assessment of his range of motion using a goniometer for accuracy. I think that the diners at Culvers were just as fascinated at what was taking place in our booth as I was watching the customers in the drive through ordering an early lunch while carrying a recently harvested buck in the back of their pickups. It is deer season in Wisconsin.
Suffice it to say that my patient was thrilled at my willingness to save him a trip to Chicago but he was even happier about the outcome in his arthritic knee. I was thrilled at his response to care. Whereas prior to the right knee intervention, he had constant pain and limitation in function, at six months, the pain and swelling are gone and he has unlimited function be it on stairs, kneeling or climbing in and out of the tractor. What separates me from the madding crowd is the data and evidence for which my recommendations are advanced. Incidentally, two blogs ago, I listed our clinical trials, the basis for our evidence based practice. Below is the trial in which my Wisconsin patient is enrolled and for which I sought follow up:
- Stem Cell Counts and the Outcome of Bone Marrow Concentrate intra-articular and intra-osseous (subchondroplasty) interventions at the knee for grades 2 and 3 OA. (supported in part by Celling). Ongoing
His results are so good that his wife decided to be next in line for her knee that did not respond well to arthroscopic surgery two years ago.
Whether you didn’t respond to surgery or haven’t had surgery, call 312 475 1893 to schedule an appointment or watch my webinar on the website www.ilcellulartherapy.com
This Thanksgiving week, let me give thanks to those who read this Blog
Tags: arthritis, bone marrow, Bone Marrow Concentrate, cellular orthopedics, Clinical Studies, Clinical Trial. Mitchell B. Sheinkop, Knee Pain Relief, Mesenchymal Stem Cell, Osteoarthritis, regenerative medicine, stem cell treatment


